 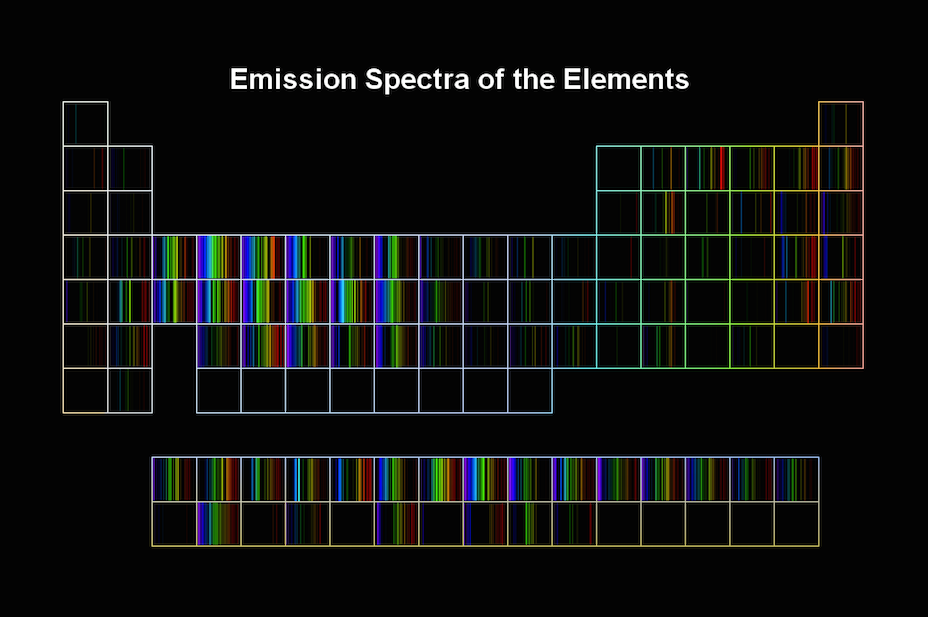 
It even contains light with a wavelength of 599.99999 nm. It contains light with a wavelength of 567.3 nm. It contains light with a wavelength of 545 nm. It contains light with a wavelength of 550 nm. Because it's continuous, that spectrum contains light with any wavelength between 500 nm and 600 nm. Suppose you have a continuous spectrum that begins with light at a wavelength of 500 nm, and ends with light at a wavelength of 600 nm. If you find that definition confusing, consider the following example. A continuous electromagnetic spectrum, then, includes every possible wavelength of light between the wavelength at the beginning of the list and the wavelength at the end. In the last section, you learned that an electromagnetic spectrum was a list of light arranged in order of increasing wavelength. Scientists don't use the word continuous to describe homework, but they do use it to describe electromagnetic spectra (spectra is just the plural word for spectrum). It means no gaps, no holes, and no breaks of any kind. When scientists use the word continuous, it has a similar meaning. Every time you finish one assignment, the teacher gives another, so you never get a break. You probably mean that the homework seems to be non-stop. 
If you have a class that requires a lot of work, you might find yourself saying something like, "I'm continuously doing homework for this class". Recognize that all elements have unique atomic spectra.Ĭontinuous Spectra Compared to Discontinuous Spectra.Recognize that white light is actually a continuous spectrum of all possible wavelengths of light.Distinguish between continuous and discontinuous spectra.To understand what light can tell us about different chemicals, though, we must first look at the electromagnetic spectrum a little more carefully. In fact, the same chemical will interact differently with differently colored beams of light. Different chemicals behave differently when struck with a beam of light. It turns out that scientists can actually learn a lot about chemicals by observing how they interact with light. What does light have to do with chemicals? Why do chemists need to know about light? Chemists are responsible for studying chemicals. Light, like matter, is part of the universe, but chemists aren't responsible for studying the entire universe. You might wonder, though, why a chemistry textbook would waste a whole lesson on light. We now know how light can act as a wave or a particle, depending on the situation. 2 Continuous Spectra Compared to Discontinuous Spectra. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
